
Founded in 1976, SOPHYSA was the pioneer in the development of adjustable neurosurgical valves to treat hydrocephalus. Introduced in 1984, the Sophy® valve was the first adjustable valve in the World allowing non-invasive adjustment of operating pressures, to match an individual patient's changing clinical needs.
Acquired in 1989 by the Japanese tkb Group, SOPHYSA expanded quickly in France, and abroad, gaining global recognition in the field of Neurosurgery.
In 2004, SOPHYSA launched the Polaris® valve, the first adjustable valve with a lock, specifically designed to resist unintentional changes in operating pressure – a milestone in adjustable valve safety. Polaris® offers the patient an unequalled security against the clinical risks associated with those dysadjustments.
In 2005, SOPHYSA introduced Pressio®,a user-friendly Intra-Cranial Pressure monitoring system for neurosurgery and neurotrauma, bringing the continuous intracranial pressure monitoring to a new level of performance and ease of use.
As a neurosurgery specialist, SOPHYSA manufactures and markets a wide range of neurosurgical implants for the treatment of hydrocephalus, including various types of valves, catheters, reservoirs and accessories, a range of ICP sensors and external drainage systems, as well as subcutaneous spinal access ports.
SOPHYSA markets its products worldwide through an international network of exclusive distributors. In France and Benelux, SOPHYSA products are marketed by direct sales representatives.
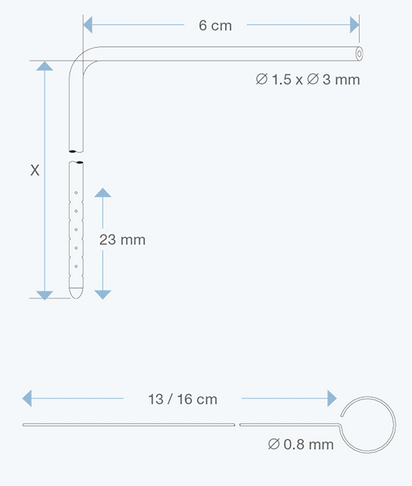
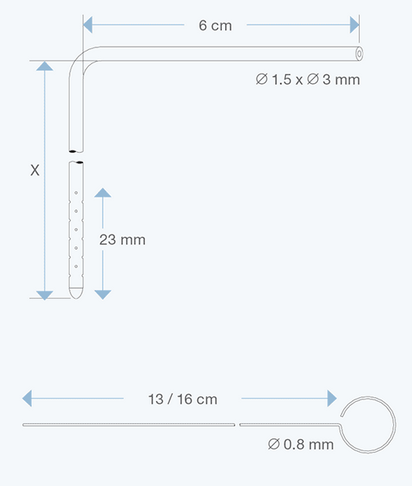
The Right Angle Ventricular Catheters, adult size, 1.5 mm ID, 3.0 mm OD, with various intracranial lengths (x in cm) and with an extracranial length of 6 cm, are multiperforated over a distance of 23 mm from the proximal tip. They are made of radiopaque silicone elastomer and include an introducing stylet.
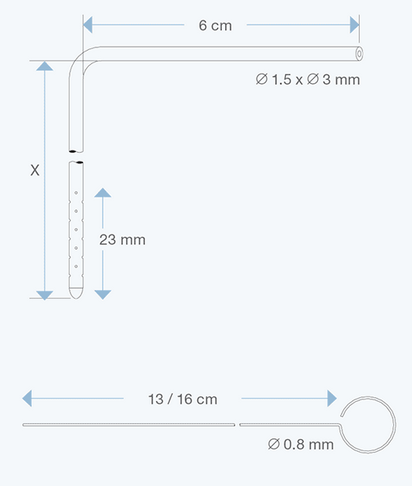
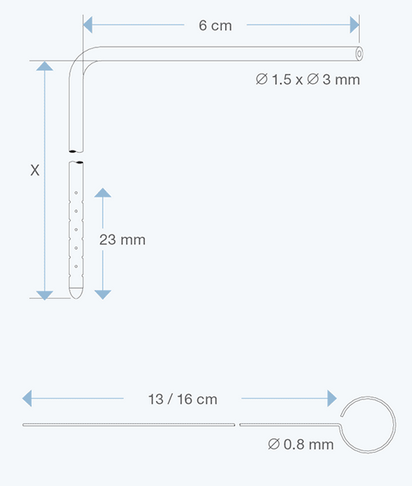
The Right Angle Ventricular Catheters, adult size, 1.5 mm ID, 3.0 mm OD, with various intracranial lengths (x in cm) and with an extracranial length of 6 cm, are multiperforated over a distance of 23 mm from the proximal tip. They are made of radiopaque silicone elastomer and include an introducing stylet.
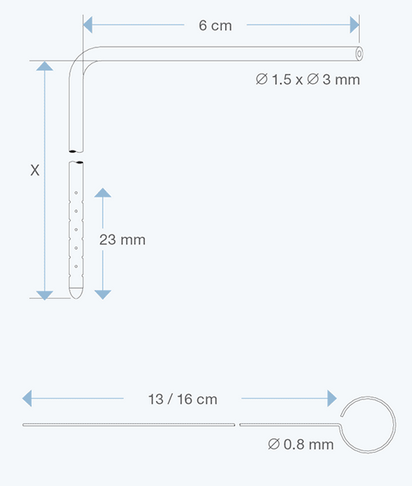
The Right Angle Ventricular Catheters, adult size, 1.5 mm ID, 3.0 mm OD, with various intracranial lengths (x in cm) and with an extracranial length of 6 cm, are multiperforated over a distance of 23 mm from the proximal tip. They are made of radiopaque silicone elastomer and include an introducing stylet.
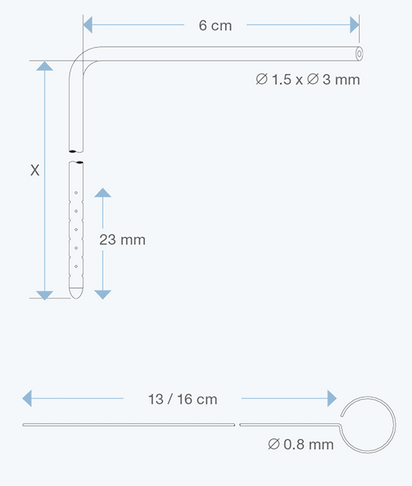
The Right Angle Ventricular Catheters, adult size, 1.5 mm ID, 3.0 mm OD, with various intracranial lengths (x in cm) and with an extracranial length of 6 cm, are multiperforated over a distance of 23 mm from the proximal tip. They are made of radiopaque silicone elastomer and include an introducing stylet.
The Right Angle Ventricular Catheters, adult size, 1.5 mm ID, 3.0 mm OD, with various intracranial lengths (x in cm) and with an extracranial length of 6 cm, are multiperforated over a distance of 23 mm from the proximal tip. They are made of radiopaque silicone elastomer and include an introducing stylet.
The Right Angle Ventricular Catheters, adult size, 1.5 mm ID, 3.0 mm OD, with various intracranial lengths (x in cm) and with an extracranial length of 6 cm, are multiperforated over a distance of 23 mm from the proximal tip. They are made of radiopaque silicone elastomer and include an introducing stylet.
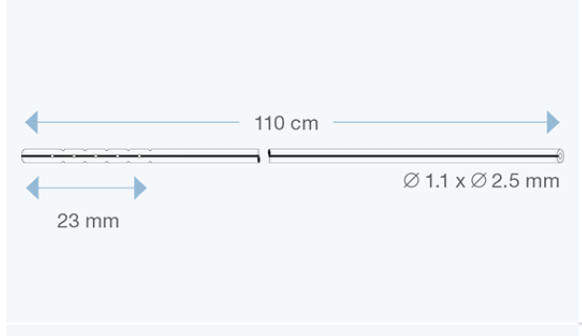
Sophysa atrial/peritoneal catheters are specifically designed for CSF drainage from the valve to the right atrium of the heart, or to the peritoneal cavity.
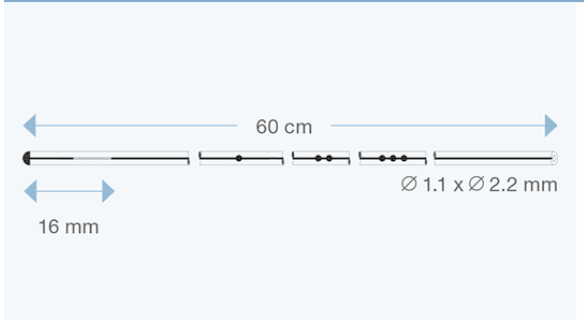
Sophysa atrial/peritoneal catheters are specifically designed for CSF drainage from the valve to the right atrium of the heart, or to the peritoneal cavity.
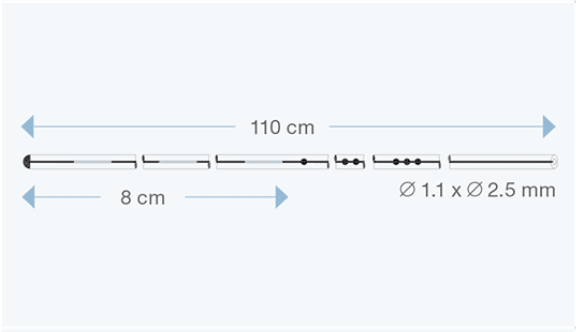
Sophysa atrial/peritoneal catheters are specifically designed for CSF drainage from the valve to the right atrium of the heart, or to the peritoneal cavity.

The Sophysa LP550 Lumbo-Peritoneal Catheter Set is specificaly designed for CSF drainage from the subarachnoid space of the spine.
Catheters only/valve NOT included.
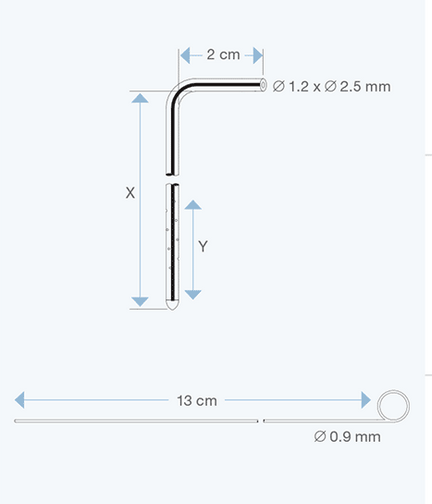
The Right Angle Ventricular Catheters, pediatric size, 1.2 mm ID, 2.5 mm OD, with various intracranial lengths (x in cm) and with an extracranial length of 2 cm, are multiperforated over a distance of 23 mm from the proximal tip. They are made of radiopaque silicone elastomer and include an introducing stylet.

The Right Angle Ventricular Catheters, pediatric size, 1.2 mm ID, 2.5 mm OD, with various intracranial lengths (x in cm) and with an extracranial length of 2 cm, are multiperforated over a distance of 23 mm from the proximal tip. They are made of radiopaque silicone elastomer and include an introducing stylet.

The Right Angle Ventricular Catheters, pediatric size, 1.2 mm ID, 2.5 mm OD, with various intracranial lengths (x in cm) and with an extracranial length of 2 cm, are multiperforated over a distance of 23 mm from the proximal tip. They are made of radiopaque silicone elastomer and include an introducing stylet.

The Right Angle Ventricular Catheters, pediatric size, 1.2 mm ID, 2.5 mm OD, with various intracranial lengths (x in cm) and with an extracranial length of 2 cm, are multiperforated over a distance of 23 mm from the proximal tip. They are made of radiopaque silicone elastomer and include an introducing stylet.

The Right Angle Ventricular Catheters, pediatric size, 1.2 mm ID, 2.5 mm OD, with various intracranial lengths (x in cm) and with an extracranial length of 2 cm, are multiperforated over a distance of 23 mm from the proximal tip. They are made of radiopaque silicone elastomer and include an introducing stylet.

Sophysa atrial/peritoneal catheters are specifically designed for CSF drainage from the valve to the right atrium of the heart, or to the peritoneal cavity.
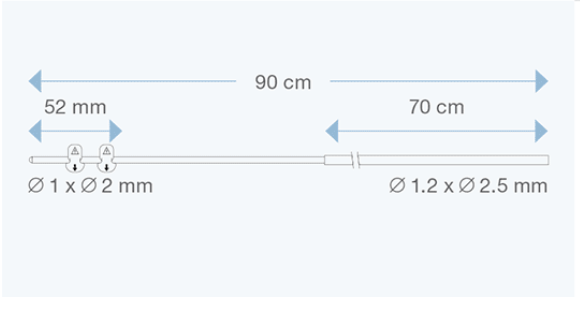
Sophysa atrial/peritoneal catheters are specifically designed for CSF drainage from the valve to the right atrium of the heart, or to the peritoneal cavity.
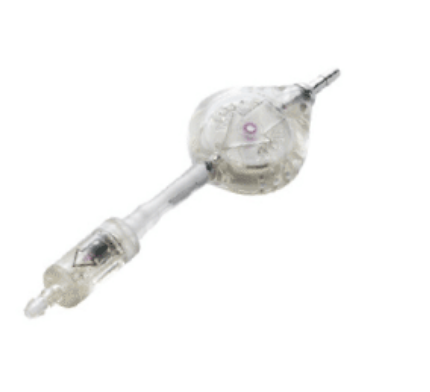
The device incorporates a ball-in-cone mechanism, well known for its precision and reliability.
It is placed downstream of a CSF shunting valve for the treatment of hydrocephalus, giving it additional resistance depending on the position of the patient. This makes it possible to limit the siphon effect during CSF drainage when the patient stands up.
A tantalum weight ball presses on a ruby ball, which occludes the aperture for the passage of the CSF.
When SiphonX® is in the vertical position, the ruby ball is subjected to the full weight of the tantalum ball, occludes the anti-siphon aperture and the device adds 200 mmH 2O to the operating pressure of the valve.
When SiphonX®is in the horizontal position, the ruby ball is not subjected to the weight of the tantalum ball and so does not occlude the aperture of the anti-siphon device.
In this horizontal position, the device is therefore open and does not add any additional resistance to the operating pressure of the valve.
For all intermediate positions, SiphonX® adds a resistance which depends on the angle of inclination.
By design SiphonX®is not affected by the implantation height relatively to cerebral ventricules.

The device incorporates a ball-in-cone mechanism, well known for its precision and reliability.
It is placed downstream of a CSF shunting valve for the treatment of hydrocephalus, giving it additional resistance depending on the position of the patient. This makes it possible to limit the siphon effect during CSF drainage when the patient stands up.
A tantalum weight ball presses on a ruby ball, which occludes the aperture for the passage of the CSF.
When SiphonX® is in the vertical position, the ruby ball is subjected to the full weight of the tantalum ball, occludes the anti-siphon aperture and the device adds 200 mmH 2O to the operating pressure of the valve.
When SiphonX®is in the horizontal position, the ruby ball is not subjected to the weight of the tantalum ball and so does not occlude the aperture of the anti-siphon device.
In this horizontal position, the device is therefore open and does not add any additional resistance to the operating pressure of the valve.
For all intermediate positions, SiphonX® adds a resistance which depends on the angle of inclination.
By design SiphonX®is not affected by the implantation height relatively to cerebral ventricules.
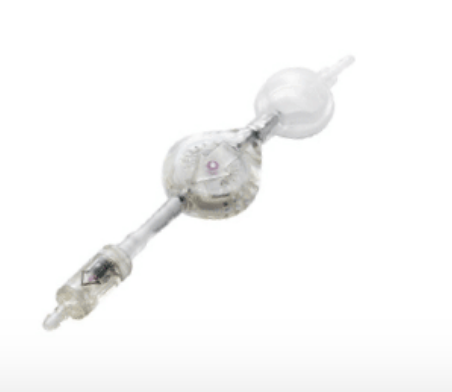
The device incorporates a ball-in-cone mechanism, well known for its precision and reliability.
It is placed downstream of a CSF shunting valve for the treatment of hydrocephalus, giving it additional resistance depending on the position of the patient. This makes it possible to limit the siphon effect during CSF drainage when the patient stands up.
A tantalum weight ball presses on a ruby ball, which occludes the aperture for the passage of the CSF.
When SiphonX® is in the vertical position, the ruby ball is subjected to the full weight of the tantalum ball, occludes the anti-siphon aperture and the device adds 200 mmH 2O to the operating pressure of the valve.
When SiphonX®is in the horizontal position, the ruby ball is not subjected to the weight of the tantalum ball and so does not occlude the aperture of the anti-siphon device.
In this horizontal position, the device is therefore open and does not add any additional resistance to the operating pressure of the valve.
For all intermediate positions, SiphonX® adds a resistance which depends on the angle of inclination.
By design SiphonX®is not affected by the implantation height relatively to cerebral ventricules.

The device incorporates a ball-in-cone mechanism, well known for its precision and reliability.
It is placed downstream of a CSF shunting valve for the treatment of hydrocephalus, giving it additional resistance depending on the position of the patient. This makes it possible to limit the siphon effect during CSF drainage when the patient stands up.
A tantalum weight ball presses on a ruby ball, which occludes the aperture for the passage of the CSF.
When SiphonX® is in the vertical position, the ruby ball is subjected to the full weight of the tantalum ball, occludes the anti-siphon aperture and the device adds 200 mmH 2O to the operating pressure of the valve.
When SiphonX®is in the horizontal position, the ruby ball is not subjected to the weight of the tantalum ball and so does not occlude the aperture of the anti-siphon device.
In this horizontal position, the device is therefore open and does not add any additional resistance to the operating pressure of the valve.
For all intermediate positions, SiphonX® adds a resistance which depends on the angle of inclination.
By design SiphonX®is not affected by the implantation height relatively to cerebral ventricules.